Synthetic Data in Healthcare Market Overview
The Global Synthetic Data in Healthcare Market reached US$500.32 Million in 2024, rising to US$657.92 Million in 2025, and is expected to reach US$5882.74 Million by 2033, growing at a CAGR of 31.5% from 2026 to 2033.
The global Synthetic Data in Healthcare Market is anchored in the rapid expansion of digital health, healthcare AI, and privacy-sensitive data use across clinical and research settings. In the United States, nearly 96% of non-federal acute care hospitals have adopted certified electronic health records, creating a strong foundation of digitized clinical data for analytics and secondary use. In parallel, 71% of U.S. hospitals reported using predictive AI integrated into their EHRs in 2024, highlighting the growing need for large, high-quality datasets for model development and validation. Growth is also supported by expanding digital health ecosystems in emerging markets; in India, more than 73.98 crore ABHA IDs and over 49.06 crore linked health records had been created as of February 6, 2025. Together, these trends are increasing demand for synthetic data to support privacy-preserving analytics, AI training, clinical research, and secure healthcare data sharing.
Synthetic Data in Healthcare Market Industry Trends and Strategic Insights
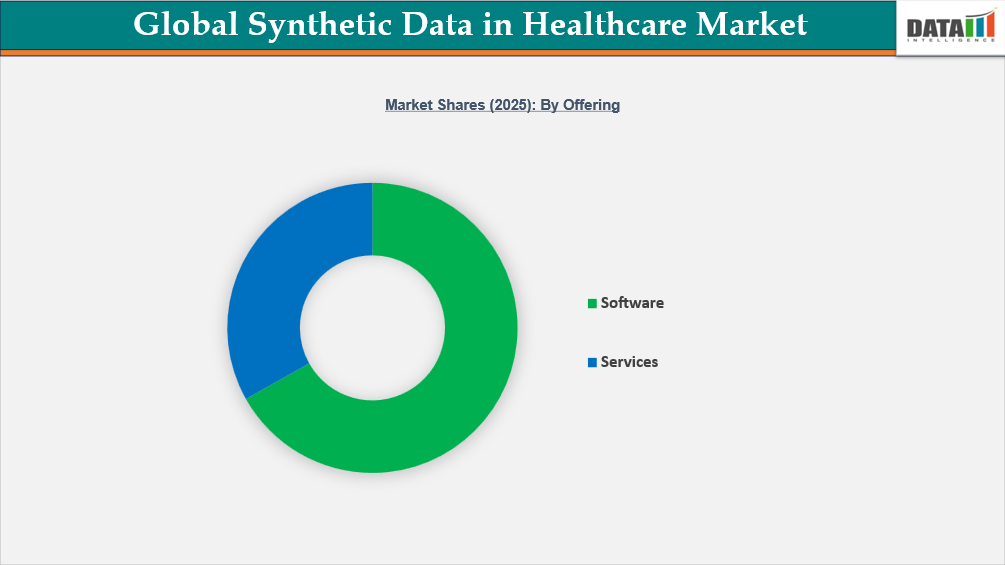
- North America leads the Global Synthetic Data in Healthcare Market, capturing the largest revenue share of 44.8% in 2025.
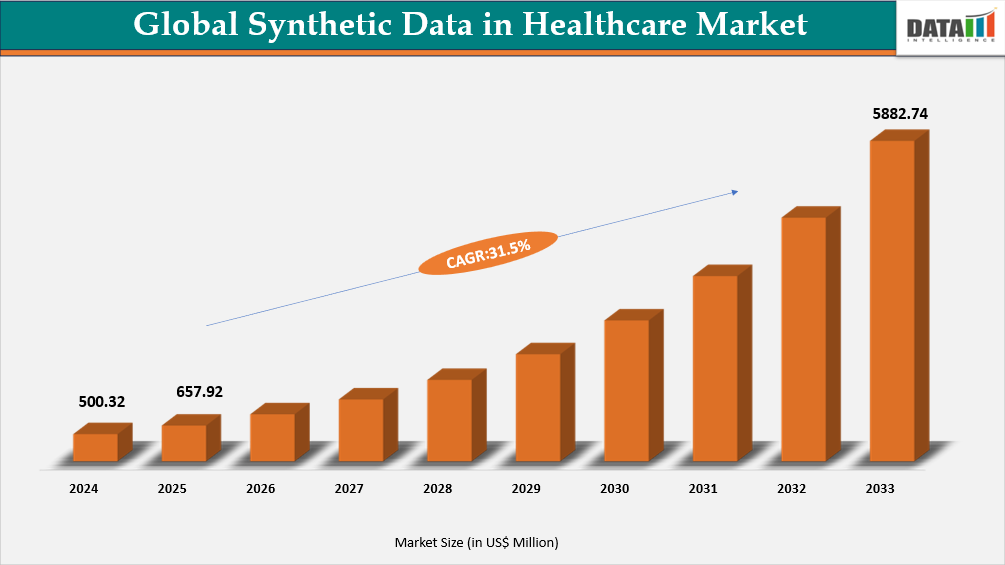
- By Offering segment, Software led the Global Synthetic Data in Healthcare Market, capturing the largest revenue share of 66.84% in 2025.
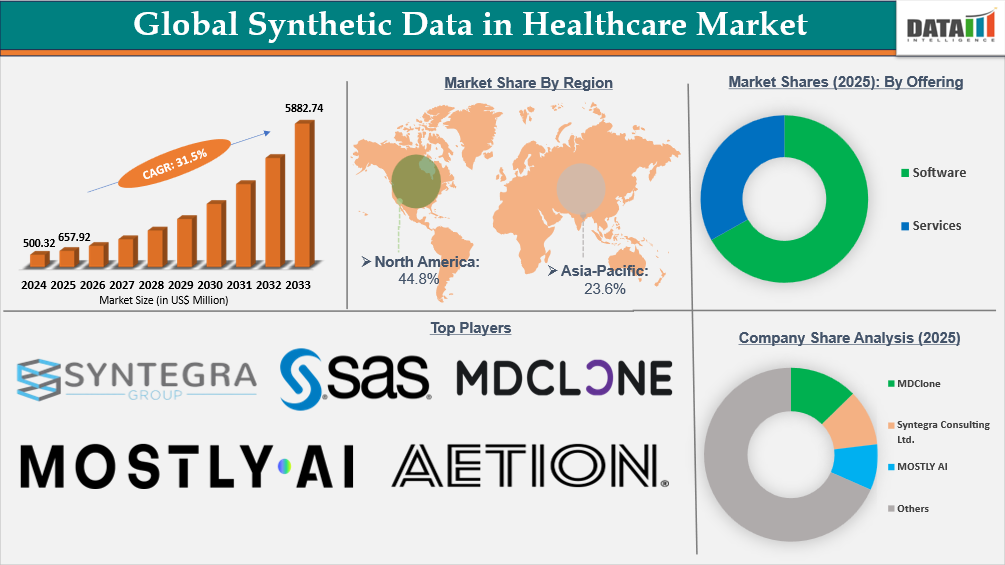
Global Synthetic Data in Healthcare Market Size and Future Outlook
- 2025 Market Size: US$657.92 Million
- 2033 Projected Market Size: US$5882.74 Million
- CAGR (2026–2033):31.5%
- Dominating Market: North America
- Fastest Growing Market: Asia-Pacific
Market Dynamics
Increasing Use of Synthetic Data in Clinical Trials and Drug Development
The growing adoption of synthetic data in clinical trials and drug development is a key driver of the global synthetic data in healthcare market. It helps pharmaceutical and healthcare companies address challenges related to limited access to real-world patient data, privacy restrictions, and delays in study execution. Traditional clinical trials often face enrollment difficulties, with nearly 80% of trials failing to meet enrollment timelines, increasing the need for alternative data approaches. Synthetic data supports trial simulation, protocol optimization, external control arm development, and AI-based research by generating realistic datasets without exposing sensitive health information. Its increasing relevance in rare disease research, precision medicine, and AI-enabled drug discovery is further strengthening its role as a strategic tool for faster, more efficient, and privacy-compliant healthcare innovation.
Segmentation Analysis
The Global Synthetic Data in Healthcare Market is segmented based on offering, deployment, data type, application, end user, and region.
Rising Adoption of Software Platforms in the Global Synthetic Data in Healthcare Market
Software is the core offering in the Global Synthetic Data in Healthcare Market because it provides healthcare organizations with the infrastructure to generate, de-identify, validate, and operationalize synthetic datasets for research, AI development, software testing, and secure data sharing. These platforms enable the creation of synthetic versions of patient-level data, such as EHRs, claims records, clinical notes, and imaging-linked datasets, while maintaining the statistical characteristics of the original data and minimizing privacy risks. According to the U.S. Department of Health and Human Services, synthetic data supports several key healthcare use cases, including simulation and prediction research, algorithm and methods testing, epidemiology and public health studies, health IT development, education and training, public dataset release, and data linkage.
The segment is gaining strategic importance as healthcare and life sciences organizations move synthetic data from experimental initiatives into large-scale operational workflows. In a March 2025 SAS study, 46% of healthcare entities and 56% of life sciences organizations reported that they were already using synthetic data or actively considering its adoption, indicating strong demand for platforms that enable compliant AI development and analytics. At the same time, advancements in the field are expanding platform capabilities beyond structured datasets. In October 2025, Philips highlighted that synthetic medical imaging can address training-data shortages, enhance AI model robustness, and protect patient privacy, while requiring rigorous validation to ensure quality, privacy protection, and clinical relevance. These developments demonstrate how software platforms are evolving toward more advanced multimodal healthcare data environments.
Geographical Penetration
Largest Market:
Demand for Synthetic Data in Healthcare Market in North America
Demand for synthetic data in the North American healthcare market is growing significantly due to the region’s advanced digital health infrastructure and increasing adoption of artificial intelligence in healthcare research. In the United States, approximately 96% of non-federal acute care hospitals have adopted certified electronic health record (EHR) systems, creating vast volumes of digital health data that require privacy-preserving solutions for analysis and sharing. At the same time, the use of AI in healthcare is expanding rapidly, with the U.S. FDA approving over 690 AI/ML-enabled medical devices as of 2023, highlighting the growing reliance on large datasets for model training and validation. Synthetic data is increasingly being used to overcome patient data privacy restrictions under regulations such as HIPAA while enabling secure data sharing for clinical research, drug development, and healthcare analytics. These factors are driving strong demand for synthetic data solutions across healthcare providers, pharmaceutical companies, and research institutions in North America.
U.S. Synthetic Data in Healthcare Market Outlook
The U.S. synthetic data in healthcare market is expected to witness strong growth, supported by the country’s highly digitized healthcare ecosystem, increasing AI adoption, and strict data privacy requirements. In 2024, 71% of U.S. non-federal acute care hospitals reported using predictive AI integrated into their EHRs, up from 66% in 2023, reflecting rising demand for large, high-quality datasets for model development and validation. In parallel, 70% of U.S. non-federal acute care hospitals engaged in all four domains of interoperable electronic health information exchange in 2023, showing the growing volume and movement of digital clinical data across the system. The market is also supported by a strict privacy environment, as HHS notes that HIPAA’s de-identification standard requires covered entities to use either Expert Determination or Safe Harbor methods when handling health information for secondary use. In addition, the FDA said in January 2025 that it had authorized more than 1,000 AI-enabled devices through established premarket pathways, further increasing demand for privacy-preserving data solutions such as synthetic data.
Canada Synthetic Data in Healthcare Market Trends
Canada’s synthetic data in healthcare market is trending upward as the country pushes for more connected digital health systems while still facing data-sharing and privacy constraints. In 2023, only 35% of Canadians reported accessing their own electronic health information in the past two years, below the international survey average of 45%, while in 2024, only 29% of physicians in Canada reported sharing patient clinical summaries electronically with providers outside their practice. These gaps in interoperability and data access are increasing the need for privacy-preserving tools such as synthetic data to support analytics, AI model training, and collaborative research without exposing patient identities. At the same time, Health Canada’s Pan-Canadian AI for Health Guiding Principles, published on January 30, 2025, place strong emphasis on privacy, security, representative data, and responsible AI adoption, which supports longer-term demand for high-quality synthetic healthcare datasets.
Fastest Growing Market:
Asia-Pacific Records the Fastest Growth in Synthetic Data in Healthcare Market
Asia-Pacific is expected to record the fastest growth in the synthetic data in healthcare market, driven by the region’s rapid healthcare digitization, expanding AI adoption, and massive growth in structured patient data. The region’s healthcare systems span 27 countries and territories in the OECD-WHO Asia/Pacific framework, highlighting a broad and increasingly digital healthcare base. In India, the Ayushman Bharat Digital Mission has created more than 73.98 crore ABHA IDs and linked over 49.06 crore health records as of 2025, reflecting the sharp expansion of interoperable digital health data. In Singapore, the Ministry of Health said it is injecting about S$200 million over five years into the MOH Health Innovation Fund to support innovations in public healthcare institutions, including AI. These developments are accelerating demand for privacy-preserving, scalable, and high-quality data environments, positioning Asia-Pacific as the fastest-growing regional market for synthetic data in healthcare.
India Synthetic Data in Healthcare Market Insights
India is witnessing growing momentum in synthetic data adoption in healthcare due to the rapid expansion of artificial intelligence research, large patient datasets, and government initiatives promoting digital health innovation. The country is emerging as a global hub for AI development, with over 3,000 AI startups operating in India as of 2024, many of which are actively working on healthcare analytics, medical imaging, and predictive diagnostics. In addition, India’s healthcare system generates massive volumes of patient data due to its large population of over 1.4 billion people, creating significant demand for privacy-preserving data environments that allow secure data sharing for research and AI model training. As healthcare providers, pharmaceutical companies, and health-tech startups increasingly adopt AI-driven solutions, synthetic data is gaining importance as a tool to support model development while ensuring compliance with emerging data protection regulations.
China Synthetic Data in Healthcare Market Industry Growth
China’s synthetic data in healthcare market is expected to witness strong growth, driven by the country’s massive healthcare system, rapid AI adoption, and increasing digital health infrastructure. By the end of 2024, China had about 1.09 million medical and health institutions, including around 39,000 hospitals, supported by nearly 12.95 million medical technical personnel and more than 10.37 million hospital beds. This vast healthcare network generates enormous volumes of clinical and patient data, creating strong demand for privacy-preserving technologies such as synthetic data for analytics, research, and AI model training.
In addition, China is rapidly expanding its artificial intelligence ecosystem. As of August 2024, more than 190 generative AI services had been registered in the country with over 600 million registered users, reflecting the scale of AI adoption across industries, including healthcare. The combination of large-scale healthcare data generation, strong government support for AI development, and growing demand for secure data-sharing frameworks is accelerating the adoption of synthetic data solutions in China’s healthcare sector..
Competitive Landscape
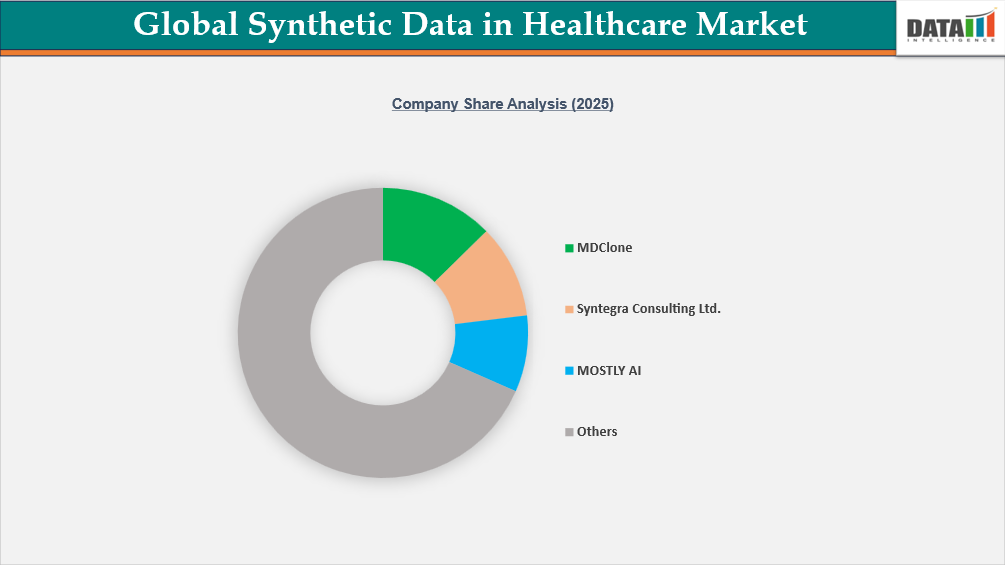
The global synthetic data in healthcare market is highly competitive, led by specialized synthetic data and privacy-enhancing technology providers such as Syntegra Consulting Ltd., MDClone, MOSTLY AI, Tonic AI, Inc., Statice, Synthesis HealthSoft, Aetion, Inc., SAS Institute Inc., Anonos, and Duality. These companies are strengthening their market positions by offering platforms that enable privacy-preserving data generation, secure data sharing, AI model development, clinical research support, and healthcare analytics across hospitals, life sciences companies, payers, and research institutions.
Competition in the market is driven by the rising demand for compliant healthcare data access, the growing use of AI and machine learning in clinical and operational workflows, and the increasing need for secure collaboration without exposing sensitive patient information. Market participants are focusing on product innovation, privacy-enhancing technologies, improvements in synthetic data quality, and integration with healthcare data ecosystems to differentiate their offerings. Strategic partnerships, platform enhancements, and expansion into real-world evidence, clinical research, and digital health applications continue to shape the competitive landscape, encouraging continuous innovation and broader adoption across the healthcare sector.
Key Developments
- In January 2026, synthetic data platforms further expanded their support for unstructured enterprise data, with Tonic.ai introducing its Unstructured Data Catalog. This advancement reflects the growing industry focus on managing text-heavy data environments, enabling more effective use of unstructured healthcare records and supporting AI-driven workflows across clinical and operational settings.
- In October 2025, synthetic medical imaging emerged as a major advancement in the healthcare synthetic data landscape. Philips highlighted the use of synthetic medical images to enhance the robustness and performance of artificial intelligence models used in healthcare applications. These synthetic imaging datasets help address training-data shortages, improve model generalization, and reduce bias while protecting patient privacy by eliminating the need for identifiable clinical images. Philips also emphasized the development of validation frameworks to ensure the quality, privacy protection, and clinical reliability of synthetic medical images before deployment in healthcare AI systems.
What Sets This Global Synthetic Data in Healthcare Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, covering global demand by offering, deployment, data type, application, end user, with region-wise analysis across North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa.
- Regulatory Intelligence – In-depth assessment of global healthcare data privacy, AI governance, and digital health regulatory frameworks impacting synthetic data adoption, including HIPAA, GDPR, EHDS, FDA guidance, Health Canada, NMPA, and India’s digital health and data protection regulations, covering data use compliance, validation expectations, cross-border data transfer requirements, and governance standards.
- Competitive Benchmarking – Structured benchmarking of leading synthetic data platform providers, healthcare AI companies, and digital health data solution vendors based on technology capabilities, data fidelity, privacy-preserving features, healthcare use cases, geographic presence, partnerships, and innovation strategies across clinical research, medical imaging, and healthcare analytics.
- Geographic & Emerging Market Coverage – Regional analysis highlighting digital health maturity, AI adoption in healthcare, regulatory readiness, interoperability progress, and demand for privacy-preserving data solutions, with special focus on high-growth opportunities across North America, Europe, Asia-Pacific, Latin America, and the Middle East.
- Actionable Strategies & Cost Dynamics – Regional analysis highlighting digital health maturity, AI adoption in healthcare, regulatory readiness, interoperability progress, and demand for privacy-preserving data solutions, with special focus on high-growth opportunities across North America, Europe, Asia-Pacific, Latin America, and the Middle East.