Regulatory Affairs & Compliance Market Overview
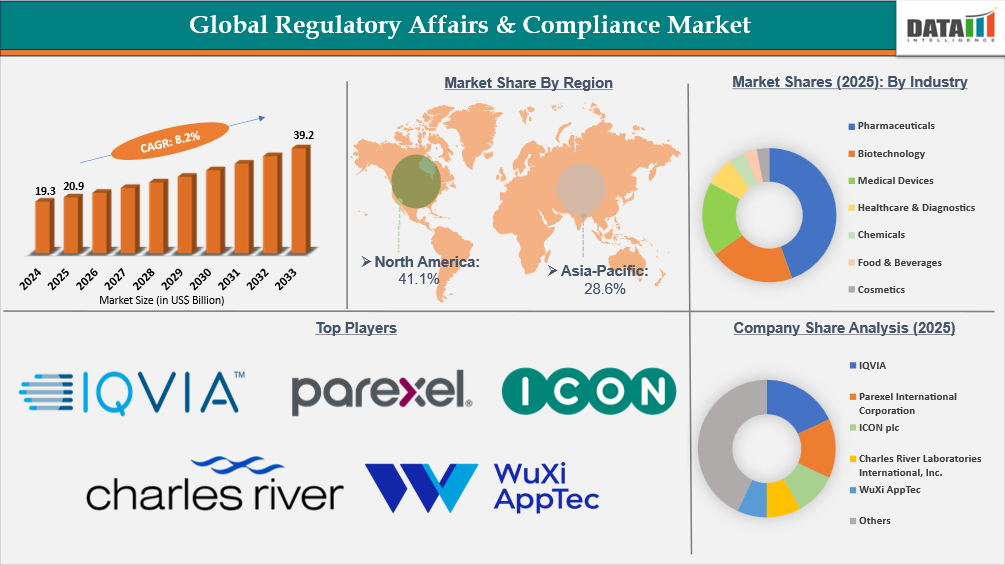
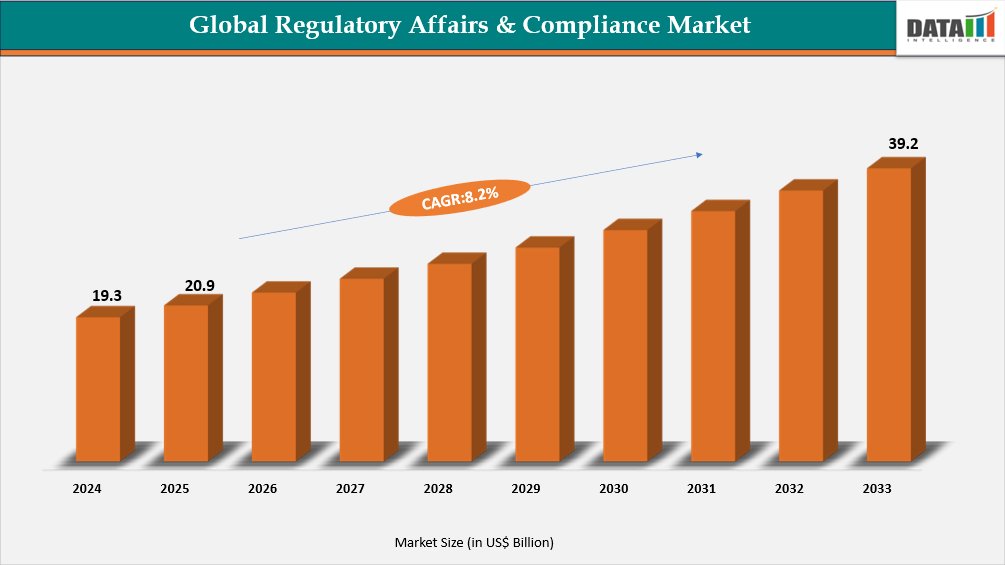
The Global Regulatory Affairs & Compliance market reached US$19.3 Billion in 2024, rising to US$20.9 Billion in 2025 and is expected to reach US$39.2 Billion by 2033, growing at a CAGR of 8.2% from 2026 to 2033.
The market is witnessing strong expansion due to increasing regulatory complexity across the pharmaceutical, biotechnology, medical device, and healthcare industries. Rising global investments in drug discovery, growing clinical trial activities, and the surge in approvals for innovative therapies are significantly increasing the need for regulatory strategy, submission management, and compliance monitoring services. Regulatory authorities across major regions, including the FDA, EMA, NMPA, PMDA, and CDSCO, are continuously strengthening regulatory standards, increasing compliance requirements for life science companies operating across multiple geographies.
Additionally, the growing adoption of biologics, biosimilars, gene therapies, and personalized medicine is increasing regulatory documentation and approval complexity. The globalization of pharmaceutical supply chains and cross-border commercialization of healthcare products is further driving demand for harmonized regulatory submissions and global compliance strategies. Technological advancements are also transforming the market, with increasing adoption of artificial intelligence, regulatory information management systems (RIMS), automation tools, and digital compliance platforms improving regulatory workflows and reducing approval timelines. Furthermore, increasing outsourcing of regulatory affairs and compliance functions by pharmaceutical and biotechnology companies is supporting market growth as organizations seek cost efficiency and specialized regulatory expertise.
Regulatory Affairs & Compliance Industry Trends and Strategic Insights
- North America leads the global regulatory affairs & compliance market, capturing the largest revenue share of 41.1% in 2025.
- By industry, pharmaceuticals led the global regulatory affairs & compliance market, capturing the largest revenue share of 44.49% in 2025.
Global Regulatory Affairs & Compliance Market Size and Future Outlook
- 2025 Market Size: US$20.9 Billion
- 2033 Projected Market Size: US$39.2 Billion
- CAGR (2026–2033): 8.2%
- Dominating Market: North America
- Fastest Growing Market: Asia-Pacific
For More detailed information Request for Sample
Market Dynamics
Growth in Pharmaceutical R&D, Clinical Trials, and New Drug Approvals
The continuous growth in pharmaceutical R&D, increasing clinical trial activities, and rising approvals of new drugs are significantly driving demand for regulatory affairs and compliance services worldwide. Pharmaceutical and biotechnology companies are expanding their innovation pipelines, resulting in a higher number of investigational products entering clinical development and requiring extensive regulatory documentation, submission management, and adherence to evolving guidelines from major regulatory authorities such as the U.S. FDA, EMA, and Pharmaceuticals and Medical Devices Agency. According to the U.S. Food and Drug Administration, 50 novel drugs were approved in 2024, following 55 approvals in 2023, reflecting the strong pace of pharmaceutical innovation and regulatory review activity.
Additionally, around 66% of therapies approved in 2024 were reviewed under expedited regulatory programs, indicating the growing complexity of regulatory pathways and accelerated approval mechanisms. As the number of clinical trials, regulatory submissions, and post-approval compliance requirements continues to increase globally, pharmaceutical companies are increasingly relying on specialized regulatory consulting and outsourcing providers to manage submissions, ensure global compliance, and accelerate product approvals, thereby strengthening the growth of the global regulatory affairs and compliance market.
Segmentation Analysis
The global regulatory affairs & compliance market is segmented based on service type, category, industry, deployment mode, regulatory lifecycle stage, regulatory authority, and region.
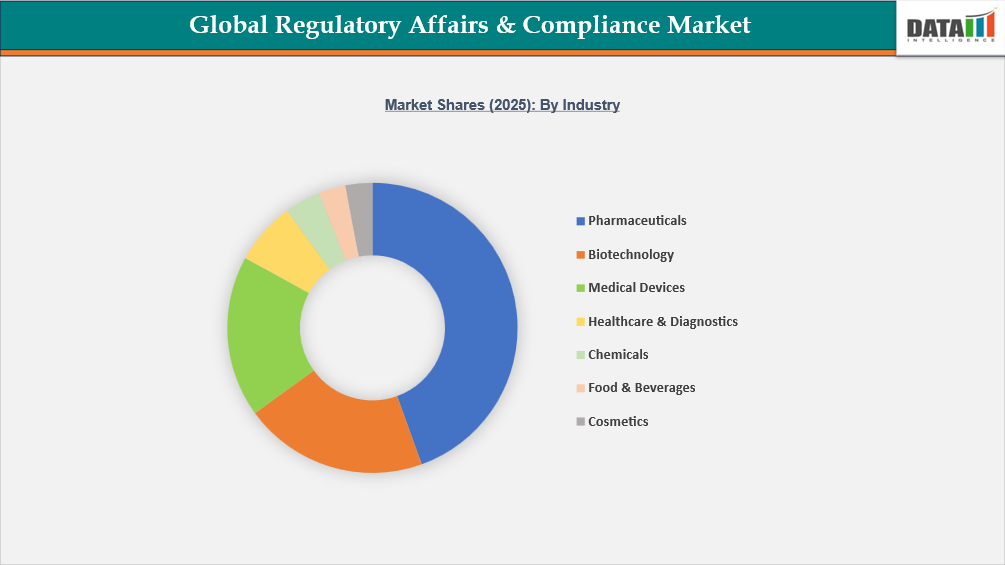
Pharmaceutical Industry Driving the Largest Demand for Regulatory Affairs & Compliance Services
The pharmaceuticals segment holds the largest share of the global regulatory affairs & compliance market, accounting for approximately 44.49% of the total market. This dominance is driven by the highly complex and strictly regulated nature of pharmaceutical product development, approval, and post-market monitoring. Drug development requires extensive regulatory documentation, clinical trial approvals, marketing authorization submissions, and continuous pharmacovigilance across multiple regulatory jurisdictions. In addition, the growing number of global clinical trials has significantly increased regulatory oversight and submission requirements. For instance, more than 5,318 new clinical trial starts were recorded globally in 2024, highlighting the rising regulatory workload associated with trial approvals, safety monitoring, and compliance reporting. Furthermore, many new therapies are evaluated through accelerated regulatory pathways such as priority review and breakthrough therapy programs, which require intensive regulatory coordination and documentation. As pharmaceutical companies increasingly commercialize products across multiple international markets, they rely heavily on specialized regulatory consulting, submission management, and compliance monitoring services, reinforcing the segment’s dominant position in the regulatory affairs and compliance market.
Geographical Penetration
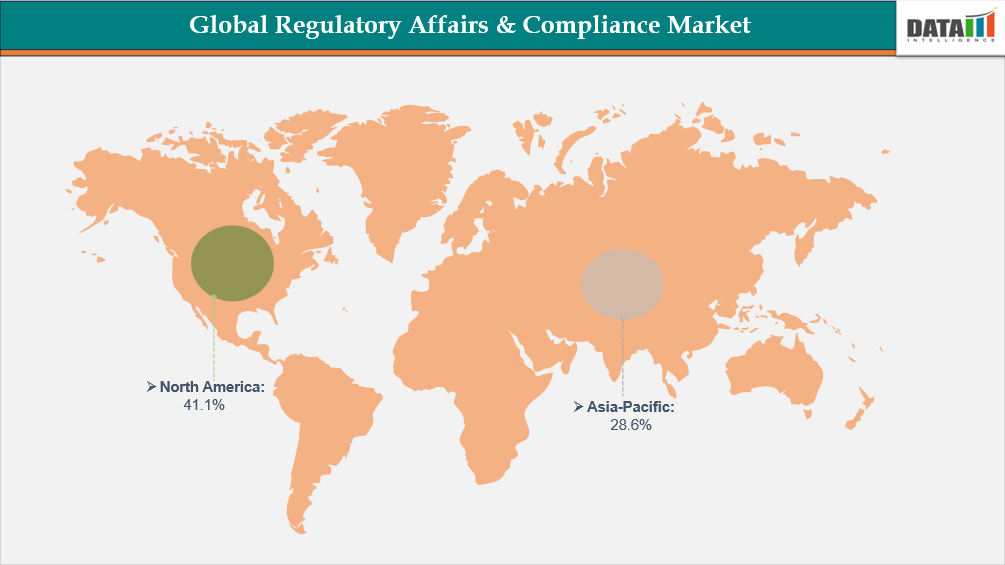
Largest Market:
Demand for Regulatory Affairs & Compliance Market in North America
North America is the largest regional market for regulatory affairs & compliance services, driven by the presence of a mature pharmaceutical and biotechnology ecosystem, stringent regulatory requirements, and high volumes of clinical trials and drug approvals. The United States and Canada are home to many of the world’s leading life science companies, which invest heavily in research and development and require strong regulatory support across the product lifecycle from regulatory submissions to post-market compliance and pharmacovigilance. Regulatory authorities such as the U.S. FDA and Health Canada enforce strict and evolving regulatory standards, increasing demand for regulatory documentation, submission management, and compliance monitoring services. Additionally, strong adoption of digital regulatory technologies and increasing outsourcing of regulatory functions to specialized service providers further strengthen North America’s dominant position in the global regulatory affairs & compliance market.
U.S. Regulatory Affairs & Compliance Market Outlook
The U.S. regulatory affairs & compliance market remains the most advanced and influential segment globally, reflecting the country’s pivotal role in pharmaceutical, biotechnology, and medical device innovation. The market is propelled by the stringent regulatory environment enforced by the U.S. FDA, which demands comprehensive regulatory documentation, strong compliance practices, and rigorous post-market surveillance. Continued growth in clinical trial activity and new drug approvals has sustained strong demand for regulatory services. In 2025, the FDA’s Center for Drug Evaluation and Research (CDER) approved 46 novel drugs, highlighting the high level of regulatory review activity that drives ongoing requirements for regulatory coordination and compliance oversight.
In addition to traditional regulatory submission support, rapid adoption of digital regulatory technologies such as regulatory information management systems (RIMS), artificial intelligence-enabled regulatory intelligence, and automated submission platforms is transforming regulatory workflows and improving compliance efficiency. Regulatory modernization initiatives aimed at streamlining review processes and harmonizing requirements with international regulators further contribute to market expansion.
Canada Regulatory Affairs & Compliance Market Trends
The Canada regulatory affairs & compliance market is experiencing steady growth driven by increasing clinical research activities, strong regulatory harmonization efforts, and rising adoption of digital compliance technologies across the life sciences sector. Regulatory authorities such as Health Canada are strengthening alignment with global regulatory frameworks like the U.S. FDA and EMA to streamline approval pathways and improve data transparency. The country is also witnessing expanding clinical trial activity, which is a key driver for regulatory submissions, safety monitoring, and compliance documentation. According to Clinical Trials Ontario and national research data, Canada accounts for more than 3,000 active clinical trials in 2025, reflecting strong pharmaceutical and biotechnology research activity that supports demand for regulatory services. In addition, the growing focus on pharmacovigilance, post-market surveillance, and quality compliance standards is further strengthening regulatory service demand across the Canadian healthcare ecosystem.
Fastest Growing Market:
Asia-Pacific Records the Fastest Growth in the Regulatory Affairs & Compliance Market
The Asia-Pacific region is recording the fastest growth in the global regulatory affairs & compliance market, driven by the rapid expansion of pharmaceutical, biotechnology, and medical device sectors across emerging economies such as China, India, and South Korea. Increasing clinical trial activity, rising domestic manufacturing capabilities, expanding regulatory frameworks, and growing investments in research and development have significantly amplified demand for regulatory strategy, submissions management, compliance monitoring, and regulatory intelligence services in the region.
Regulatory authorities in major Asia-Pacific markets are also strengthening alignment with international standards, which increases the complexity and volume of regulatory submissions for global product launches. The combination of strong healthcare growth, rising adoption of advanced therapies, and expanding life sciences ecosystems positions Asia-Pacific as the fastest-growing regional market for regulatory affairs and compliance services worldwide.
India Regulatory Affairs & Compliance Market Insights
The India regulatory affairs & compliance market is emerging as a significant growth region within the broader Asia-Pacific landscape, driven by expanding pharmaceutical and biotechnology activities, increasing clinical research, and strengthening regulatory frameworks. India has become a key hub for clinical trials, generics manufacturing, and biotechnology innovation, which is increasing demand for regulatory strategy, submission management, compliance monitoring, and post-market regulatory functions. Industry data indicates that approximately 7,455 clinical trials were registered in India in 2024, reflecting the country’s growing role in global clinical research and the increasing regulatory requirements for trial approvals, safety reporting, and compliance documentation.
Additionally, Indian life sciences companies are increasingly outsourcing regulatory and compliance services to improve operational efficiency, support global market expansion, and adopt digital regulatory technologies such as regulatory information management systems and automated submission platforms. These factors collectively position India as a rapidly expanding market for regulatory affairs and compliance services in the Asia-Pacific region.
China Regulatory Affairs & Compliance Market Industry Growth
The China regulatory affairs & compliance market is experiencing robust growth driven by the rapid expansion of the pharmaceutical, biotechnology, and medical device industries, along with increasing participation in global life sciences innovation. China is one of the fastest-growing markets for regulatory services due to strong investments in pharmaceutical research and development, expansion of advanced therapy pipelines, and rising clinical research activities. Chinese regulatory authorities, particularly the National Medical Products Administration (NMPA), have introduced regulatory reforms to accelerate drug and device approvals, improve regulatory transparency, and align local regulatory standards with international frameworks, thereby increasing demand for regulatory strategy, submission management, and compliance monitoring services. Growing globalization of Chinese pharmaceutical exports and increasing partnerships with multinational companies are further strengthening the need for global regulatory intelligence, harmonized submission platforms, and end-to-end compliance solutions.
Competitive Landscape
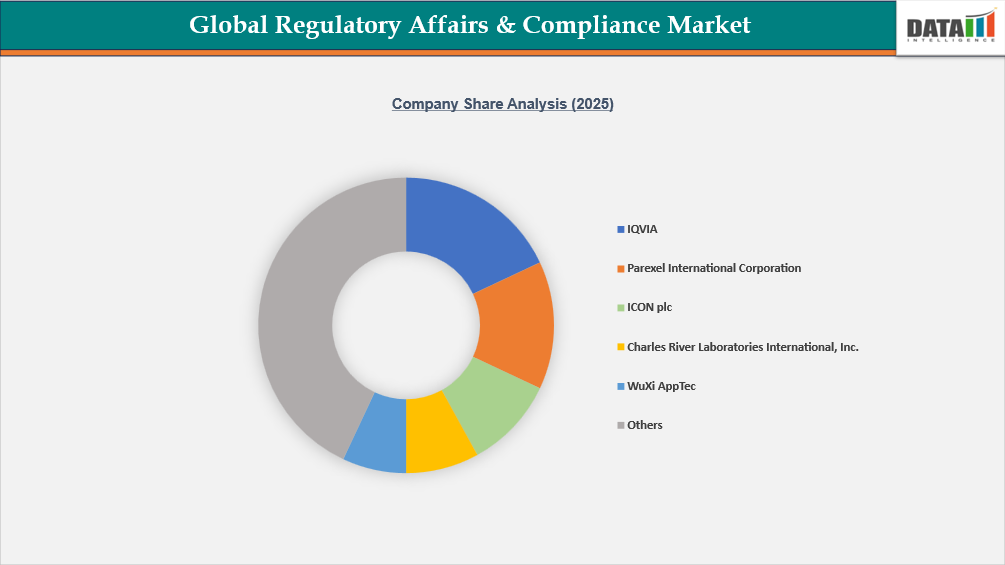
The global regulatory affairs & compliance market is highly competitive, with several established contract research organizations (CROs), regulatory consulting firms, and professional service providers offering comprehensive regulatory strategy, submission management, and compliance support. Key companies operating in this market include Parexel International (MA) Corporation, ICON plc, IQVIA, WuXi AppTec, Charles River Laboratories International, Inc., Labcorp, and Medpace, which leverage their global regulatory expertise, clinical research capabilities, and integrated service portfolios to support pharmaceutical, biotechnology, and medical device companies throughout the product development lifecycle. Additionally, specialized regulatory service providers such as Freyr, ProPharma Group MIS Limited, and PHARMALEX GMBH focus on regulatory submissions, pharmacovigilance, and lifecycle management services.
Meanwhile, large consulting and digital transformation firms including Genpact, Cognizant, Accenture, and Deloitte, are strengthening their presence by delivering regulatory technology solutions, compliance consulting, and digital regulatory intelligence platforms. Companies in this market increasingly focus on strategic partnerships, mergers and acquisitions, and digital regulatory platforms to enhance global submission capabilities and support faster regulatory approvals in increasingly complex regulatory environments.
Key Developments
- In September, 2025, Parexel International (MA) Corporation announced a strategic partnership with Weave Bio to integrate artificial intelligence into regulatory workflows. The collaboration focuses on automating regulatory documentation and accelerating submission preparation, helping pharmaceutical and biotechnology companies streamline approval timelines and improve regulatory efficiency.
- In February 2025, Freyr Solutions launched a new “Market Intel” service designed to help life-science companies track global regulatory changes, competitor strategies, and compliance risks. The solution provides real-time regulatory updates and analytics to support regulatory planning and global market entry strategies.
What Sets This Global Regulatory Affairs & Compliance Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, covering global demand for regulatory affairs and compliance services segmented by service type, category, industry, deployment mode, regulatory lifecycle stage, and regulatory authority, along with detailed regional analysis across North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa.
- Regulatory Intelligence – In-depth assessment of global regulatory frameworks affecting pharmaceutical, biotechnology, and medical device industries, including requirements from regulatory authorities, including FDA, EMA, NMPA, PMDA, and CDSCO, covering regulatory submission pathways, compliance standards, and post-marketing surveillance requirements.
- Competitive Benchmarking – Structured benchmarking of leading regulatory consulting firms, CROs, and professional service providers based on regulatory expertise, service portfolios, geographic presence, digital regulatory capabilities, and strategic partnerships with pharmaceutical and biotechnology companies.
- Geographic & Emerging Market Coverage – Regional analysis highlighting regulatory environment complexity, clinical trial activity, pharmaceutical R&D investments, and demand for regulatory compliance services, with special focus on growth opportunities in Asia-Pacific, Latin America, and Middle Eastern markets.
- Actionable Strategies & Cost Dynamics – Strategic insights into regulatory outsourcing trends, digital regulatory transformation, cost optimization strategies, and lifecycle regulatory management, supported by expert perspectives from regulatory consultants, pharmaceutical companies, and industry stakeholders.
























































