Japan Pharmaceuticals Advanced Aseptic Processing Market Overview
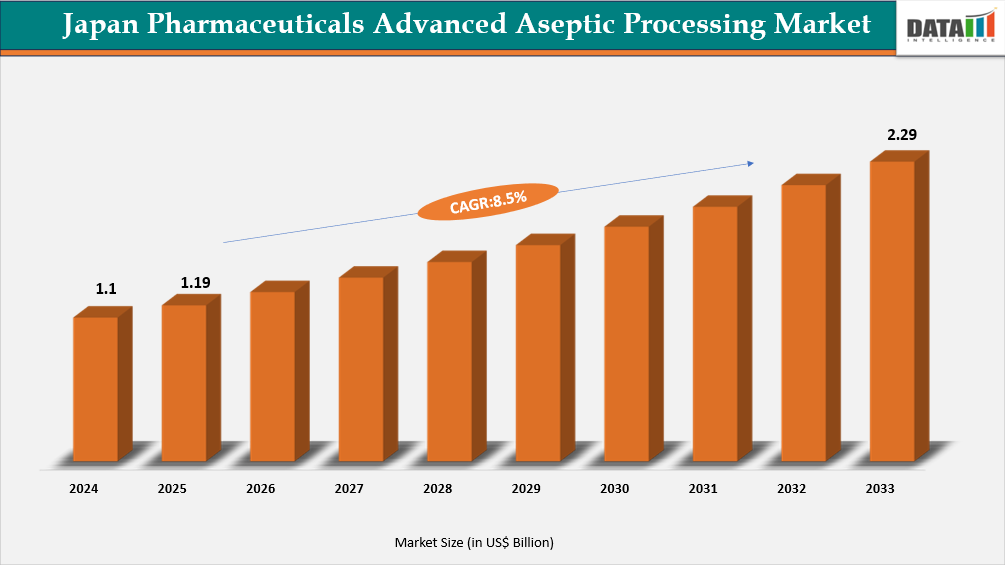
The Japan Pharmaceuticals Advanced Aseptic Processing market reached US$1.1 billion in 2024, rising to US$1.19 billion in 2025 and is expected to reach US$2.29 billion by 2033, growing at a CAGR of 8.5% from 2026 to 2033.
Japan's advanced aseptic processing market is driven by a unique combination of solid healthcare infrastructure, a large elderly population, and a high prevalence of chronic and complicated illnesses, all of which contribute to significant demand for sterile injectable medicines. The country's 1.6 million hospital beds enable broad use of injectable biologics, chemotherapeutics, and supportive care medications. 29% of the population will be 65 or older, leading to higher rates of cancer, cardiovascular problems, and metabolic conditions requiring parenteral therapy. Japan has approximately 1 million new cancer cases yearly, highlighting the need for high-quality sterile drugs. To satisfy these needs, hospitals and manufacturers are increasingly relying on advanced aseptic technology such as isolators, closed-system filling lines, automated environmental monitoring, and robots to maintain product sterility and reduce contamination risk in high-volume manufacturing. In addition to these technical forces, Japan's national health insurance system and stable payment mechanisms encourage the use of sterile generics and biosimilars, allowing for more patient access while also improving cost efficiency. Together, these characteristics lay the groundwork for long-term and strategic growth in the country's sophisticated aseptic processing sector.
Pharmaceuticals Advanced Aseptic Processing Industry Trends and Strategic Insights
- By product type segment, injectables led the Japan Pharmaceuticals Advanced Aseptic Processing market, capturing the largest revenue share of 35% in 2025.
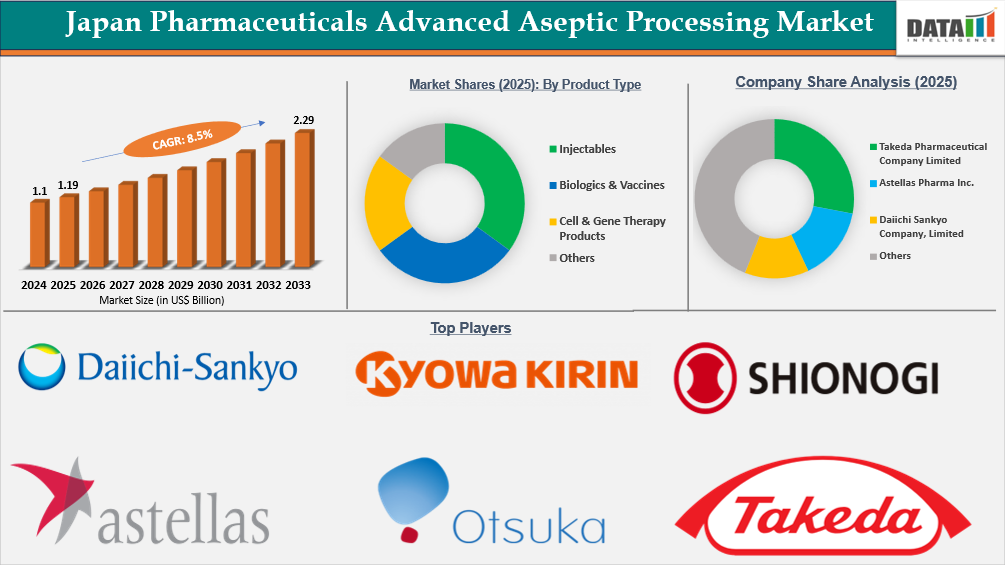
Japan Pharmaceuticals Advanced Aseptic Processing Market Size and Future Outlook
- 2025 Market Size: US$1.19 Billion
- 2033 Projected Market Size: US$2.29 Billion
- CAGR (2026–2033): 8.5%
For More Detailed Information, Request for Sample
Market Dynamics
Increasing Global Collaboration
Increasing worldwide collaboration is a major driver of the Japan Pharmaceuticals Advanced Aseptic Processing Market. Japanese pharmaceutical and biotechnology companies are increasingly collaborating with global CDMOs, technology providers, and research organizations to gain access to cutting-edge aseptic processing technologies such as robotic filling systems, barrier isolators, and single-use bioprocessing solutions. These agreements enable speedier development and commercialization of sterile biologics, vaccines, and innovative medicines while adhering to rigorous GMP guidelines. Partnerships also enable knowledge transfer, the adoption of global best practices, risk sharing, and cost savings, allowing Japanese producers to economically grow high-value sterile goods and extend their position in foreign markets. As a consequence, worldwide collaboration accelerates the adoption of modern aseptic technology, enhances production quality, and solidifies Japan's position as a leading hub for sterile pharmaceutical manufacture.
Segmentation Analysis
The Japan Pharmaceuticals Advanced Aseptic Processing market is segmented based on product type, technology, end user, packaging, and therapy area.
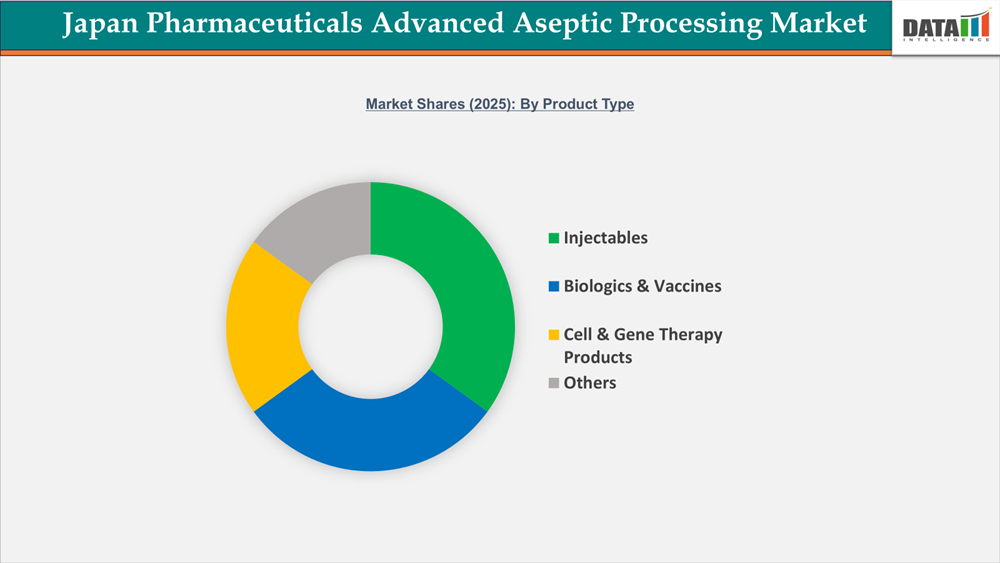
Injectables Segment Sustains Growth in Japan Pharmaceuticals Advanced Aseptic Processing Market
In the Japan Pharmaceuticals Advanced Aseptic Processing Market, injectables are sterile medicinal products delivered intravenously, intramuscularly, or subcutaneously, and they account for around 35% of the whole market. These medications are commonly utilized in hospitals and clinics for therapy such as antibiotics, cardiovascular drugs, pain relief, hormones, and anti-infectives, making them essential for patient care. Injectables must undergo extensive aseptic processing to eliminate microbiological contamination and conform with Japan's GMP and PMDA criteria, which include sterile fill-finish, filtering, lyophilization, and the use of isolators or restricted-access barrier systems. The high value and risks associated with injectables promote the use of automation and robots to eliminate human interaction, improve sterility assurance, and increase manufacturing efficiency. Companies such as Takeda, Otsuka, and Daiichi Sankyo specialize in specialized injectables and frequently work with contract manufacturers to increase sterile fill-finish capacity, addressing both hospital demand and supply chain concerns. Injectables remain a key driver of development and innovation in Japan's aseptic processing business.
Competitive Landscape
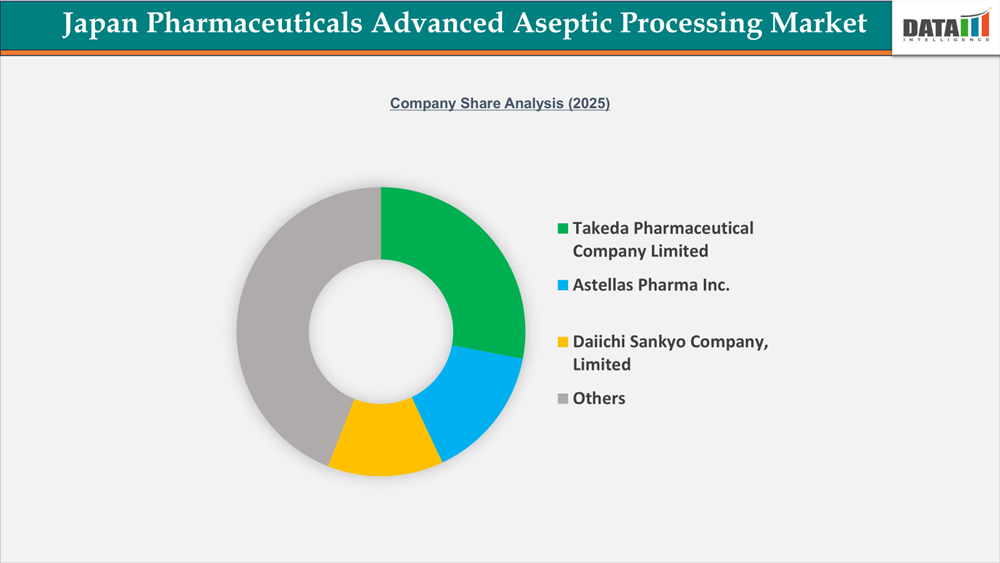
The competitive landscape of the Japan Pharmaceuticals Advanced Aseptic Processing market is moderately consolidated, characterized by the strong presence of domestic pharmaceutical leaders and specialized contract manufacturers. Companies such as Takeda Pharmaceutical Company Limited, DAIICHI SANKYO COMPANY, LIMITED., Astellas Pharma Inc., Otsuka Holdings Co., Ltd., Tanabe Pharma Corporation, and Kyowa Kirin Co., Ltd. drive the market through specialty injectables, biosimilars, and high-value hospital formulations, leveraging established regulatory expertise and distribution networks. Firms like Baxter and Fresenius Kabi India Pvt. Ltd. enhance competition by offering advanced aseptic processing capabilities, including prefilled syringes, liposomal formulations, and complex biologics. Additionally, niche players such as Shionogi & Co., Ltd. and Meiji Seika Pharma Co., Ltd. focus on oncology and anti-infective injectables, addressing the rising demand from Japan’s aging population and hospital procurement programs. Strategic collaborations, technology licensing, and expansion of aseptic portfolios in depot injections, liposomal drugs, and biosimilar injectables remain key strategies shaping competitive dynamics in this market.
Key Developments
- In March 2025, Astellas Pharma Inc. and YASKAWA Electric Corporation formed a joint venture to develop an advanced cell therapy manufacturing platform using robotics. Leveraging YASKAWA’s dual‑arm robot “Maholo,” the collaboration enhances precision, reproducibility, and automation in cell‑based and complex biologic production. This initiative reflects trends in Japan’s Pharmaceuticals Advanced Aseptic Processing Market toward automated, high‑precision sterile manufacturing, addressing production complexity, improving scalability, and supporting long-term growth in biologics.
What Sets This Japan Pharmaceuticals Advanced Aseptic Processing Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, covering Japan demand by key segmentation, with detailed analysis.
- Regulatory Intelligence – In-depth assessment of Japan pharmaceutical regulatory frameworks impacting market development and commercialization, including FDA, EMA, NMPA, PMDA, and CDSCO requirements, labeling standards, patent exclusivity, and post-marketing surveillance.
- Competitive Benchmarking – Structured benchmarking on product portfolios, pipeline strength, geographic reach, pricing, clinical differentiation, and strategic partnerships.
- Actionable Strategies & Cost Dynamics – Provides strategic insights on lifecycle management, generic entry risks, combination therapy positioning, pricing pressures, and manufacturing cost structures, informed by perspectives from regulatory advisors, pharmaceutical executives, and industry specialists.