Japan Microbiome Profiling Diagnostics Market Overview
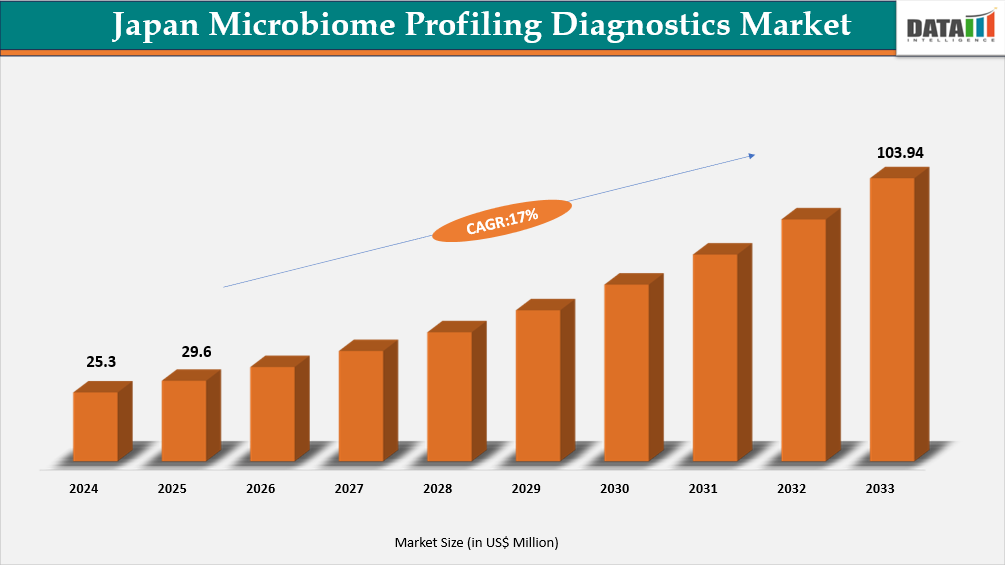
The Japan Microbiome Profiling Diagnostics Market reached US$25.3 Million in 2024, rising to US$29.6 Million in 2025 and is expected to reach US$103.94 Million by 2033, growing at a CAGR of 17% from 2026 to 2033.
Market expansion is driven by strong demand from hospitals, specialty clinics, and research institutions, where microbiome profiling is increasingly integrated into precision medicine, chronic disease management, and preventive healthcare programs. Adoption is supported by advanced sequencing platforms, bioinformatics tools, and well-established domestic laboratory infrastructure, enabling accurate diagnostics and actionable insights.
Japan’s aging population, with nearly 29% of residents aged 65 or older, contributes to a higher prevalence of metabolic, gastrointestinal, and age-related diseases, further fueling demand for microbiome-based diagnostics. In addition, rising public awareness of gut health and government initiatives promoting personalized medicine encourage broader adoption across clinical and preventive settings. The presence of the Japan Microbiome Database, which houses thousands of microbial profiles linked to lifestyle, diet, and health metadata, strengthens research capabilities and clinical application of microbiome diagnostics.
Overall, these demographic, technological, and institutional factors are expected to sustain long-term growth, positioning microbiome profiling diagnostics as a high-potential segment within Japan’s clinical diagnostics landscape.
Microbiome Profiling Diagnostics Market Industry Trends and Strategic Insights
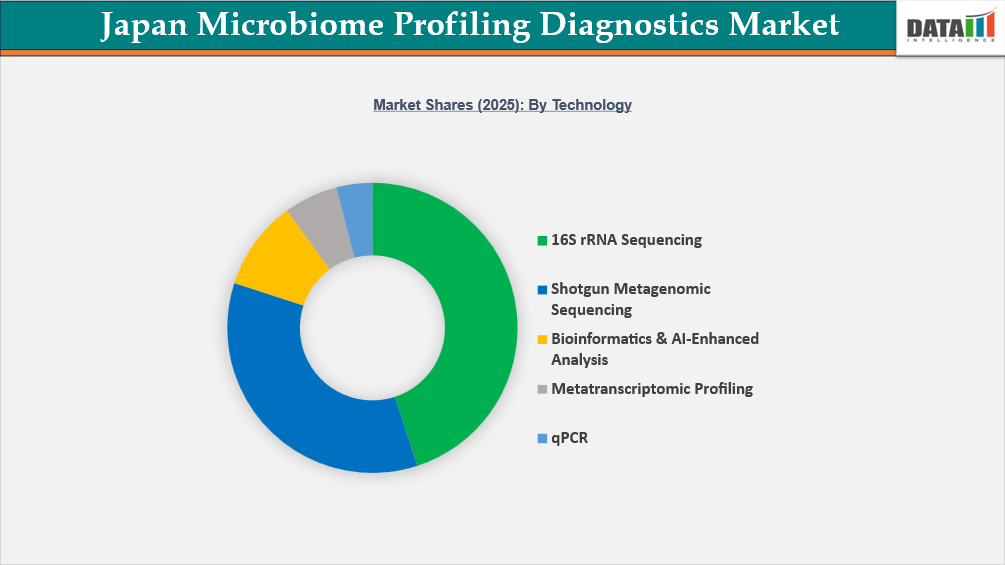
- By technology segment, 16S rRNA Sequencing led the Japan Microbiome Profiling Diagnostics Market, capturing the largest revenue share of 45% in 2025.
Japan Microbiome Profiling Diagnostics Market Size and Future Outlook
- 2025 Market Size: US$29.6 Million
- 2033 Projected Market Size: US$103.94 Million
- CAGR (2026–2033): 17%
For More Detailed Information, Request for Sample
Market Dynamics
Rapid Adoption of Next-Generation Sequencing
The rapid adoption of Next‑Generation Sequencing (NGS) is a key driver of the Japan microbiome profiling diagnostics market, as it enables highly precise and comprehensive analysis of complex microbial communities in human samples. Unlike traditional culture-based methods, NGS can simultaneously identify thousands of microbial species, providing detailed insights into microbial composition, diversity, and function. The increasing use of NGS in research and clinical settings across Japan, combined with declining costs and faster turnaround times, has made microbiome profiling more accessible and commercially viable. Moreover, the technology supports personalized medicine by allowing clinicians to tailor treatments based on individual microbiome signatures, particularly in areas such as gastroenterology, metabolic disorders, and oncology. Strong biotechnology infrastructure and supportive regulatory initiatives further facilitate the integration of NGS into diagnostic workflows, collectively driving the growth of the microbiome profiling diagnostics market in Japan.
Segmentation Analysis
The Japan Microbiome Profiling Diagnostics Market is segmented based on clinical focus, application, technology, sample type, end-user, and service.
16S rRNA Sequencing Supports Rising Demand for Japan Microbiome Profiling Diagnostics Market
In Japan, 16S rRNA sequencing is a widely used method for microbial community profiling, particularly in studies of the human gut microbiome. Culture-independent approaches have shown that up to 60–80% of gut bacterial species are difficult to culture, which has driven the extensive adoption of sequencing methods over traditional microbiology techniques. Large-scale cohort studies have demonstrated the utility of 16S rRNA gene amplicon sequencing, such as profiling the gut microbiota of 192 Japanese volunteers to benchmark primer sets for fecal microbiome analysis. Applications of 16S sequencing extend beyond the gut; for instance, it has been used to characterize salivary microbiota in Japanese oral cancer patients, identifying changes in microbial diversity and genus-level abundance associated with disease. The National Institute of Biomedical Innovation’s Japan Microbiome Database maintains thousands of 16S rRNA gene amplicon profiles from multiple Japanese cohorts, highlighting its role as a foundational resource for population-level studies. While more comprehensive approaches, such as shotgun metagenomics, provide deeper functional insights, 16S rRNA sequencing remains a primary tool for bacterial community composition and diversity profiling due to its high throughput, efficiency, and scalability across large sample sets.
Competitive Landscape
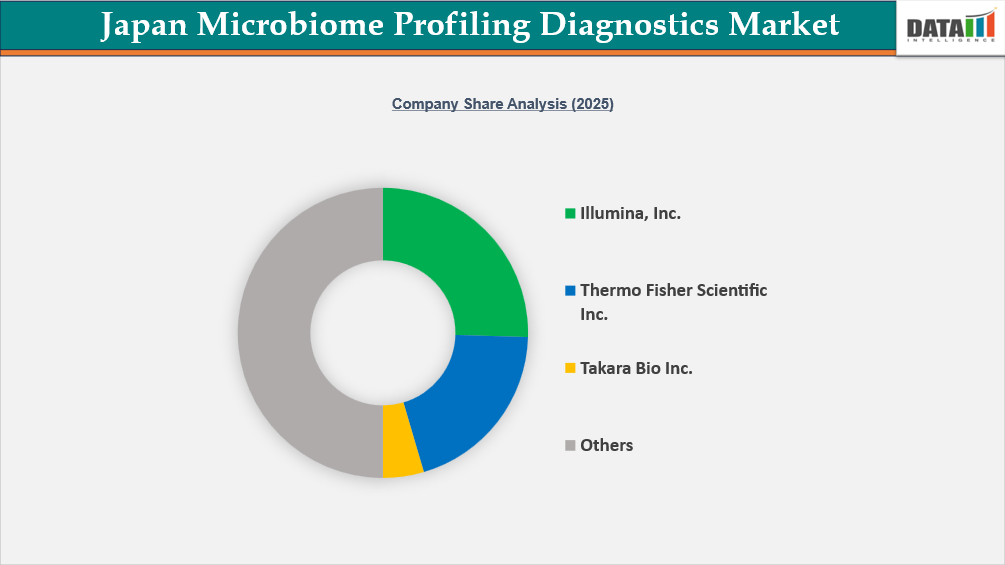
The Japan Microbiome Profiling Diagnostics Market is moderately consolidated, led by global sequencing and life sciences companies such as Thermo Fisher Scientific Inc. and Illumina, Inc., which dominate through advanced NGS platforms, reagents, and integrated bioinformatics solutions. Domestic players, including Takara Bio Inc. and Eiken Chemical Co., Ltd., leverage strong local regulatory expertise and distribution networks, focusing on PCR technologies and molecular diagnostic kits.
Specialized microbiome and longevity-focused firms such as Metagen, Inc., Zymo Research Corporation, Microba, Microbiome Insights, Genetic Analysis, and Decode Age compete through advanced gut microbiome sequencing, biological age testing, and AI-driven bioinformatics platforms, with partnerships and clinical-grade metagenomics expansion shaping the competitive landscape.
Key Developments
- In August 2025, Japan-based Metagen, Inc. announced the expansion of its personalized gut microbiome analysis services powered by its proprietary Metabologenomics® platform, strengthening its clinical and preventive healthcare positioning within the Japanese market.
What Sets This Japan Microbiome Profiling Diagnostics Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, covering Japan demand by key segmentation, with other detailed analysis.
- Regulatory Intelligence – Overview of Japan’s PMDA approval process, clinical validation requirements, labeling rules, and post-market compliance for microbiome diagnostics.
- Competitive Benchmarking – Benchmarking of key players based on technology platforms, service offerings, partnerships, and innovation strength in Japan.
- Actionable Strategies & Cost Dynamics – Insights into pricing trends, sequencing cost dynamics, reimbursement outlook, and growth strategies shaping the market.