Japan CRISPR-Cas9 Gene Editing Market Overview
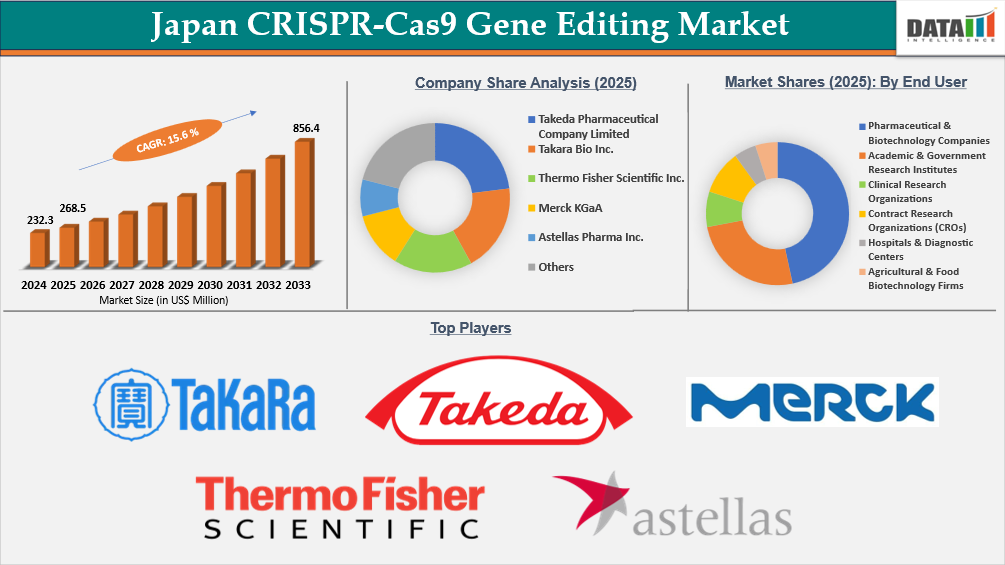
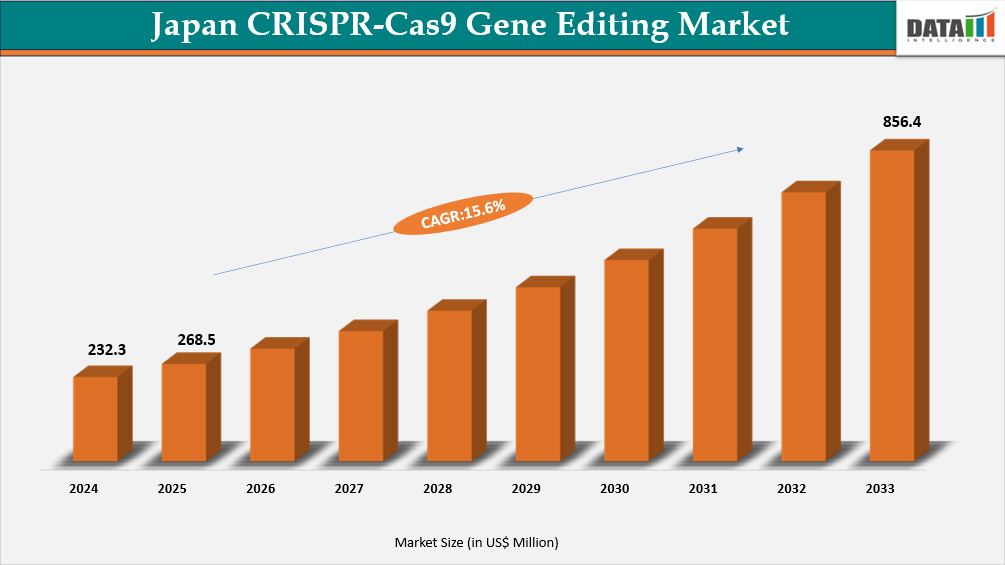
The Japan CRISPR-Cas9 gene editing market reached US$232.3 Million in 2024, rising to US$268.5 Million in 2025 and is expected to reach US$856.4 Million by 2033, growing at a CAGR of 15.6% from 2026 to 2033.
The Japan CRISPR-Cas9 gene editing market is a rapidly growing segment of the country’s biotechnology and precision medicine ecosystem, supported by strong research infrastructure, rising healthcare innovation investments, and increasing demand for advanced therapeutic solutions. CRISPR-Cas9 technology is widely applied in drug discovery, functional genomics, disease modeling, and personalized medicine, making it a critical driver of innovation in Japan’s healthcare and life sciences sector. The country’s strong academic research base and collaborations between pharmaceutical companies, biotechnology firms, and research hospitals are accelerating the clinical translation of genome editing technologies into therapeutic applications, particularly in oncology, rare genetic disorders, and regenerative medicine.
The market is further supported by increasing patient demand for precision medicine solutions, driven by Japan’s aging population and growing prevalence of chronic diseases. Regulatory frameworks supporting regenerative and gene therapy commercialization are also helping accelerate market adoption by reducing time-to-market for innovative treatments. Despite challenges such as ethical considerations surrounding genome editing, high research and development costs, and complex patent landscapes, continuous technological advancements, strategic partnerships, and growing investment in genomic technologies are expected to sustain strong market growth in Japan.
CRISPR-Cas9 Gene Editing Market Industry Trends and Strategic Insights
- By end user, pharmaceutical & biotechnology companies led the Japan CRISPR-Cas9 gene editing market, capturing the largest revenue share of 46.6% in 2025.
Japan CRISPR-Cas9 Gene Editing Market Size and Future Outlook
- 2025 Market Size: US$268.5 Million
- 2033 Projected Market Size: US$856.4 Million
- CAGR (2026–2033): 15.6%
Market Dynamics
Increasing Investments in Precision Medicine and Genomic Research in Japan
Japan is strategically expanding investments in precision medicine and genomic research, significantly accelerating the adoption of CRISPR-Cas9 technologies across therapeutic, translational, and research applications. National programs supported by the Ministry of Health, Labour and Welfare and the Japan Agency for Medical Research and Development are driving large-scale genome sequencing initiatives, rare disease research, oncology genomics, and regenerative medicine development. Government-backed funding is strengthening infrastructure for biobanks, next-generation sequencing platforms, and advanced cell and gene therapy manufacturing facilities, creating a robust ecosystem for genome editing innovation.
Japan’s focus on personalized and stratified medicine, particularly in oncology, hematology, and inherited genetic disorders, is increasing demand for precise gene modification tools such as CRISPR-Cas9 for target validation, functional genomics, and ex vivo cell therapy development. Academic institutions, including leading national universities and research hospitals, are actively collaborating with domestic biotech companies to translate genome editing research into clinical-stage therapies. Furthermore, regulatory support for regenerative and gene-based therapies under Japan’s accelerated approval pathways is reducing time to market for innovative treatments. Collectively, sustained public funding, public-private partnerships, and infrastructure expansion are positioning Japan as a competitive hub for genome editing research in Asia, thereby driving consistent growth in the Japan CRISPR-Cas9 Gene Editing Market.
Segmentation Analysis
The Japan CRISPR-Cas9 gene editing market is segmented based on product, technology, end user, application, delivery method, and disease area.
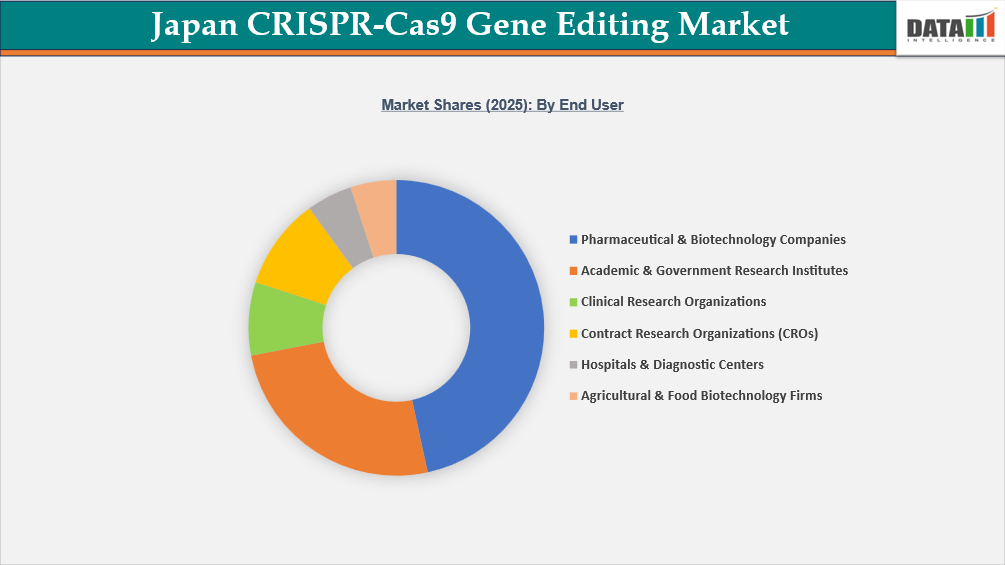
Pharmaceutical & Biotechnology Firms Hold the Largest End-User Share in Japan CRISPR-Cas9 Gene Editing Market
The pharmaceutical & biotechnology companies hold the largest share in the Japan CRISPR-Cas9 gene editing market due to their central role in therapeutic development, drug discovery, and commercialization of gene-based innovations. Japanese pharmaceutical companies are increasingly integrating CRISPR-Cas9 technologies into their R&D pipelines for oncology, rare genetic disorders, regenerative medicine, and cell and gene therapy programs. The strong innovation ecosystem supported by the Ministry of Health, Labour and Welfare, and funding initiatives from the Japan Agency for Medical Research and Development further accelerates industry adoption of advanced genome editing tools.
Unlike academic institutions that primarily focus on early-stage research, pharmaceutical and biotech companies drive large-scale commercial applications, including target validation, biomarker discovery, engineered cell therapies, and preclinical development. They also invest heavily in proprietary CRISPR platforms, licensing agreements, and strategic collaborations with universities and contract research organizations to expand therapeutic pipelines. Additionally, Japan’s supportive regulatory framework for regenerative and gene-based therapies enables faster clinical translation, encouraging companies to allocate significant capital toward CRISPR-enabled innovation. As a result, higher R&D expenditure, commercialization capability, and strong pipeline activity position pharmaceutical & biotechnology companies as the dominant end-user segment in the Japan CRISPR-Cas9 gene editing market.
Competitive Landscape
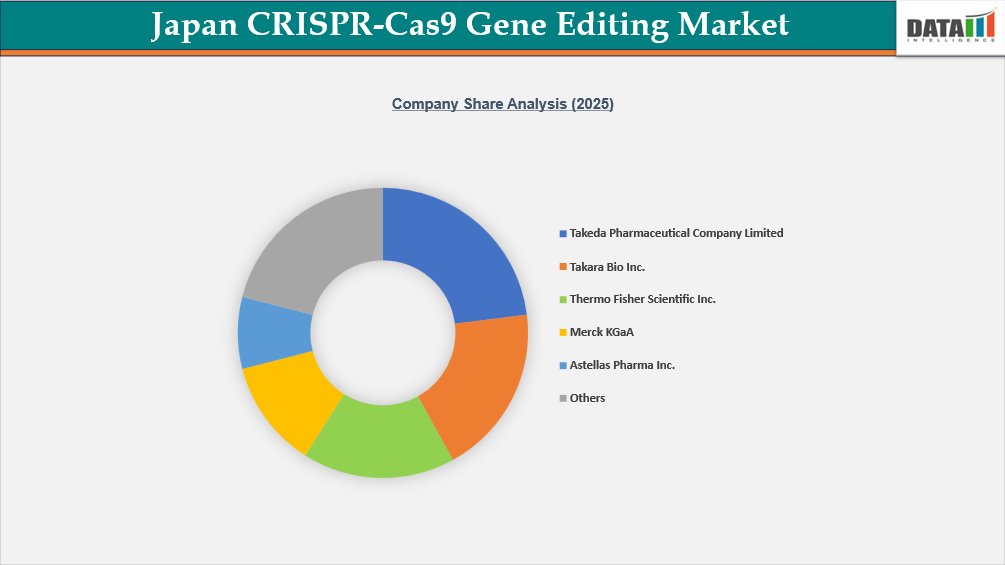
The Japan CRISPR-Cas9 gene editing market features a competitive landscape comprising leading domestic pharmaceutical companies, specialized biotechnology firms, and global life sciences technology providers. Major Japanese pharmaceutical players such as Takeda Pharmaceutical Company Limited, DAIICHI SANKYO COMPANY, LIMITED, and Astellas Pharma Inc. are leveraging CRISPR-Cas9 technologies in therapeutic research, target validation, and drug discovery, particularly in oncology and rare diseases. On the tools and reagents side, Takara Bio Inc. and Sumitomo Chemical Co., Ltd. play critical roles in supplying CRISPR kits, enzymes, guide RNAs, and oligonucleotides to academic and commercial research institutions across Japan.
Global life sciences leaders such as Thermo Fisher Scientific Inc., Merck KGaA, GenScript, and DH Life Sciences, LLC. strengthen the market through advanced CRISPR reagents, gene synthesis, and custom genome editing services. Additionally, Illumina, Inc. supports the ecosystem with next-generation sequencing platforms essential for validating gene editing outcomes. Overall, competition in Japan is driven by technological innovation, strategic collaborations, expanding therapeutic pipelines, and the growing demand for high-precision genome editing solutions.
Key Developments
- In June 2025, C4U Corporation secured up to USD 14 million (JPY 2 billion) in non-dilutive funding from the Japan Agency for Medical Research and Development under the “Strengthening Program for Pharmaceutical Startup Ecosystem” to advance its CRISPR-based hematopoietic stem cell therapy for primary immunodeficiency. The funding supports clinical validation and structured safety evaluations, including off-target assessments, reinforcing Japan’s regulatory and translational research framework. This development strengthens Japan’s genome editing ecosystem and highlights growing government-backed investment accelerating innovation within the Japan CRISPR-Cas9 Gene Editing Market.
What Sets This Japan CRISPR-Cas9 Gene Editing Market Intelligence Report Apart
- Latest Data & Forecasts – Comprehensive and up-to-date market intelligence with forecasts through 2033, segmented by product, technology, end user, application, delivery method, and disease area, with detailed Japan-level analysis.
- Regulatory Intelligence – In-depth assessment of Japan’s gene editing and regenerative medicine regulatory framework, including oversight by the Pharmaceuticals and Medical Devices Agency and the Ministry of Health, Labour and Welfare, clinical trial approval pathways, genome editing safety guidelines, patent landscape considerations, and post-marketing surveillance requirements.
- Competitive Benchmarking – Structured benchmarking of leading Japanese and global genome editing companies based on platform technology, pipeline strength, partnerships, manufacturing capabilities, and clinical differentiation within Japan.
- Actionable Strategies & Cost Dynamics – Strategic insights into commercialization pathways, government funding support, licensing trends, IP positioning, manufacturing scale-up challenges, and cost structures for ex vivo and in vivo gene editing therapies, supported by expert perspectives from biotech leaders and regulatory advisors in Japan.