Global Inflammatory Bowel Disease Market: Industry Outlook
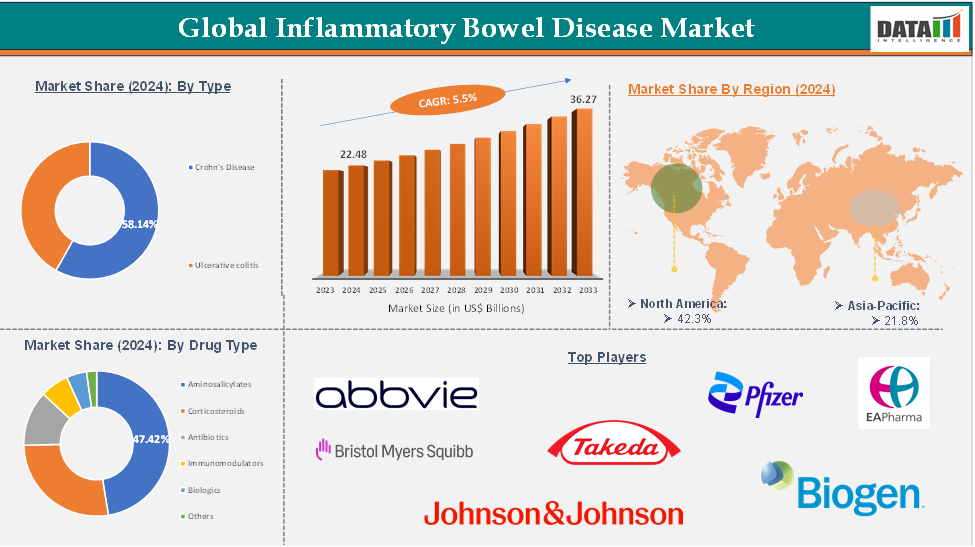
The global inflammatory bowel disease market reached US$ 22.48 Billion in 2024 and is expected to reach US$ 36.27 Billion by 2033, growing at a CAGR of 5.5% during the forecast period 2025-2033.
The global inflammatory bowel disease market is thriving due to the increasing burden of Crohn's disease and ulcerative colitis. The market is growing due to increased awareness, improved diagnostic capabilities, and the adoption of advanced therapeutics. Research and development activities support a diverse pipeline of innovative treatments. However, high costs of branded therapies and concerns over long-term safety profiles pose challenges to market expansion.
Emerging markets offer opportunities due to rising healthcare investments and urbanization. The growing availability of biosimilars makes treatments more affordable, and personalized medicine and digital health solutions are increasingly integrated into disease management strategies. The evolving treatment landscape focuses on precision therapies, microbiome-based interventions, and value-based care models, which are expected to transform patient outcomes and market dynamics.
Global Inflammatory Bowel Disease Market: Executive Summary
Global Inflammatory Bowel Disease Market Dynamics: Drivers & Restraints
Driver: Rise in prevalence of inflammatory bowel disease
The global rise in inflammatory bowel disease (IBD) is driving growth in the market, driven by factors like lifestyle changes, dietary changes, urbanization, and environmental influences. This has led to increased demand for effective treatment options, resulting in increased investment in biologics and small-molecule drugs. The growing patient base also encourages pharmaceutical companies to expand research and development efforts to address unmet clinical needs and improve long-term disease management. This trend is expected to sustain market momentum in the coming years.
For instance, according to NCBI study, the annual incidence of IBD varies by region, with estimates ranging from 10.5 to 46.14 per 100,000 in Europe, 1.37 to 1.5 per 100,000 in Asia and the Middle East, 23.67 to 39.8 per 100,000 in Oceania, 0.21 to 3.67 per 100,000 in South America, and 7.3 to 30.2 per 100,000 in North America.
Restraint: Side effects associated with the drugs
The global inflammatory bowel disease (IBD) market faces challenges due to the significant side effects of existing drug therapies. While biologics, immunosuppressants, and corticosteroids have improved IBD treatment, they are often linked to increased risks of infections, liver toxicity, malignancies, and cardiovascular complications. Long-term steroid use can lead to osteoporosis, weight gain, and metabolic disorders. These safety concerns lead to poor patient adherence, treatment discontinuation, and hesitation in aggressive therapy, limiting the widespread adoption of available treatments.
IBD drugs can cause various side effects, including mild gastrointestinal issues, fatigue, headaches, and joint pain. More serious side effects may include infections, liver problems, and an increased risk of certain cancers. Common side effects include nausea, vomiting, diarrhea, abdominal pain, bloating, fatigue, headaches, and joint pain
For more details on this report, Request for Sample
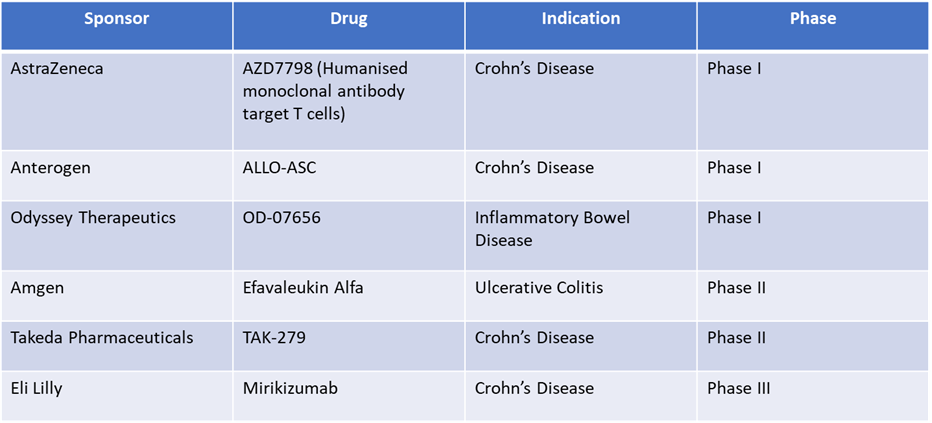
Pipeline Analysis:
Global Inflammatory Bowel Disease Market Segment Analysis
The global inflammatory bowel disease market is segmented based on type, drug type, route of administration, and region.
Product Type:
The aminosalicylates segment from the product type is expected to hold 47.42% of the inflammatory bowel disease market
Aminosalicylates are anti-inflammatory drugs used to treat inflammatory bowel diseases like ulcerative colitis and Crohn's disease. They inhibit inflammatory chemical production in the intestine, reducing inflammation and alleviating symptoms like abdominal pain, diarrhea, and rectal bleeding. Commonly used drugs include mesalamine (5-ASA), sulfasalazine, olsalazine, and balsalazide, which can be taken orally or rectally depending on inflammation severity.
The aminosalicylates segment is a significant part of the inflammatory bowel disease (IBD) market due to its clinical use, safety, and cost-effectiveness, particularly in treating mild to moderate ulcerative colitis. Globally, increasing IBD diagnosis rates and awareness among patients and healthcare providers are driving demand for first-line therapies like aminosalicylates. In developing markets, affordability and ongoing research into improved formulations support better patient compliance and outcomes.
For instance, in February 2025, Amneal Pharmaceuticals launched mesalamine 800 mg delayed-release tablets for treating moderately active ulcerative colitis in adult patients. The company also announced the launch of 5-aminosalicylic acid (5-ASA) referencing ASACOL HD and has received approval from the US Food and Drug Administration for lenalidomide capsules in various doses for treating blood cancers.
Global Inflammatory Bowel Disease Market - Geographical Analysis
North America dominated the global inflammatory bowel disease market with the highest share of 42.3% in 2024
North America dominates the global inflammatory bowel disease (IBD) market due to high disease prevalence, advanced healthcare infrastructure, and strong R&D activity. The US and Canada have the highest IBD incidence and diagnosis rates globally, with well-established diagnostic protocols and patient awareness. The region has access to advanced biologics, biosimilars, and personalized treatment options, with favorable reimbursement policies, strong regulatory support, and leading biopharmaceutical companies contributing to robust market growth. North America's leadership in IBD management and ongoing clinical research further reinforces its position as a key revenue-generating region.
For instance, in January 2025, Eli Lilly and Company received FDA approval for Omvoh, a medication used to treat Crohn's disease in adults, and two types of inflammatory bowel disease (IBD), following its approval in October 2023 as a first-in-class treatment for moderately to severely active ulcerative colitis in adults.
Asia-acific is the global Inflammatory Bowel Disease market with a market share of 21.8% in 2024
The Asia-Pacific region is experiencing significant growth in the global Inflammatory Bowel Disease (IBD) market due to rising awareness, improved diagnostic capabilities, and increasing prevalence. Factors such as urbanization, changing diets, and lifestyle-related risk factors contribute to this growth. Expanding healthcare infrastructure and access to advanced biologics and therapies are enhancing treatment outcomes. Government initiatives and pharmaceutical investments further support market growth. The region's focus on research and clinical trials is accelerating the availability of innovative treatment options, making it a vital contributor to the evolving global IBD market.
For instance, in April 2024, Takeda Pharmaceutical Company Limited received FDA approval for subcutaneous administration of Entyvio post-induction therapy, used to treat severely active Crohn's Disease patients.
Global Inflammatory Bowel Disease Market - Key Players
The major global players in the inflammatory bowel disease market include AbbVie Inc., Takeda Pharmaceutical Company Limited, Pfizer Inc., Biogen, Johnson & Johnson, Bristol-Myers Squibb Company, EA Pharma Co., Ltd, and Salix Pharmaceuticals, among others.
Global Inflammatory Bowel Disease Market – Key Developments
In September 2024, the FDA approved TREMFYA (guselkumab), a human, dual-acting monoclonal antibody targeting interleukin-23 and CD-64, for treating moderate to severely active ulcerative colitis in adult patients.
In July 2024, Teva Pharmaceuticals and Sanofi announced an update to the timing of their anti-TL1A, duvakitug program, which is investigating the human IgG1-λ2 monoclonal antibody targeting TL1A for moderate-to-severe IBD.
Global Inflammatory Bowel Disease Market: Scope
Metrics | Details | |
CAGR | 5.5% | |
Market Size Available for Years | 2022-2033 | |
Estimation Forecast Period | 2025-2033 | |
Revenue Units | Value (US$ Bn) | |
Segments Covered | Type | Crohn's Disease, Ulcerative colitis |
Drug Type | Aminosalicylates, Corticosteroids, Antibiotics, Immunomodulators, Biologics, Others | |
Route of Administration | Oral, Rectal, Subcutaneous, Intravenous | |
Regions Covered | North America, Europe, Asia-Pacific, South America, and the Middle East & Africa | |


